5-Ethynyl-dU offers convenient click conjugation with an azide to generate a label rigidly attached to one of the oligonucleotide bases. 5-Ethynyl-dU is subject to base-catalyzed hydration during cleavage and deprotection, especially when using a strong base or heat. Hydration of an ethynyl group forms a methyl ketone which subsequently blocks potential click reactions. Mild deprotection conditions are necessary when using 5-Ethynyl-dU-CE Phosphoramidite to prevent this side reaction. TIPS-5-Ethynyl-dU-CE Phosphoramidite, containing a protected alkyne, offers broader compatibility with oligonucleotide synthesis and deprotection. Protecting the 5-ethynyl group with a triisopropylsilyl (TIPS) protecting group prevents acid or base catalyzed hydration during oligonucleotide synthesis and workup. A quick treatment with TBAF removes the TIPS protecting group.
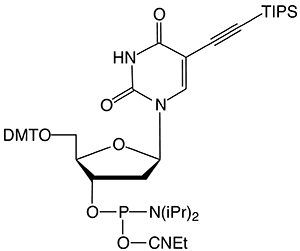
M.W.: 911.15
F.W.: 314.19
Diluent: Anhydrous Acetonitrile
Coupling: 3 minute coupling time recommended
Deprotection: No changes needed from standard method recommended by synthesizer manufacturer. TIPS removal: After deprotection, dry down oligo and take up in 0.5 mL DMF. Add 0.1 mL TBAF. Vortex and keep at 45°C for 15 minutes. Quench with 0.5 mL 2 M TEAA and desalt on Glen Gel-Pak™ column or equivalent.
Storage: Storage: Refrigerated storage, maximum of 2-8°C, dry
Stability in Solution: 1-2 Days
Please Note: All products of baseclick are patent protected and available in collaboration with baseclick. Baseclick GmbH has filed the following patent applications: 1. WO2006/117161, New labelling strategies for the sensitive detection of analytes. 2. WO2008/952775, Click Chemistry for the production of reporter molecules. Baseclick GmbH holds a worldwide license for the research market of the "Click Chemistry" patent from "The Scripps Research Institute": 3. WO03/101972, Copper-catalysed ligation of azides and acetylenes.
If you cannot find the answer to your problem then please contact us or telephone +44 (0)1954 210 200